Energy and Place Project
Essential Questions:
How does energy production and consumption impact place?
How does your sense of place, environmental ethic and understanding of our energy needs
influence your perception and decisions relating to energy production and consumption?
How does energy production and consumption impact place?
How does your sense of place, environmental ethic and understanding of our energy needs
influence your perception and decisions relating to energy production and consumption?
Through the process of researching, designing and conducting my own experiment I learned that the nature of scientific investigations are conducted in an exclusive step by step manner. Special care has to be taken during each step to ensure that an expected outcome is produced. After conducting multiple experiments throughout the year my perspective has not changed very much. But after having the opportunity to design and conduct my own I have gained insight on the amount of creativeness that is required.
After conducting my scientific investigation I was able to learn about the building blocks of the fuel I was creating. I learned how each ingredient was mixed was mixed together and the bonds that were formed to make the final biodiesel. For my particular scientific investigation, the big idea that I took away was how specific ingredients, that cannot be directly used as fuel, can be mixed and come together and form a viable alternative for fuel.
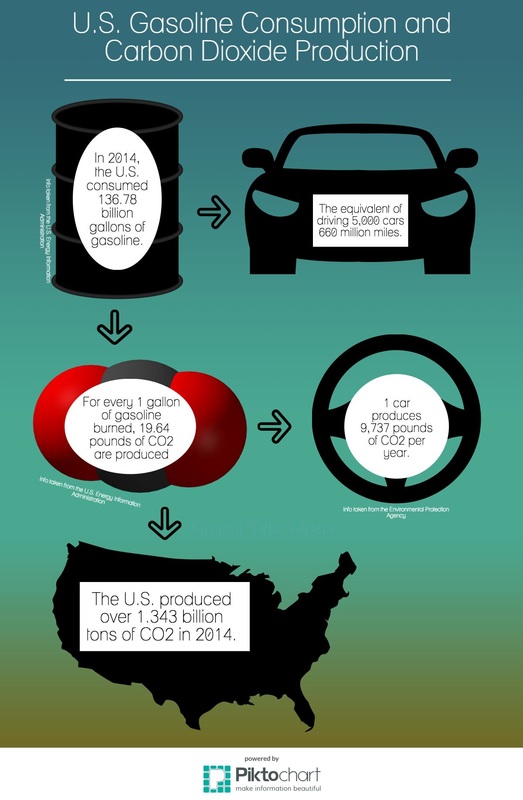
The research I did for the creation of my infographic was very informative in an overwhelming manner. I was astounded by the amount of gasoline that the United States uses and how much CO2 is emitted as a result of gasoline's consumption. Though I did not do any research pertaining to the rate gasoline depletion across the globe I was still surprised about the amount we consumed based on my knowledge of how much only one person uses.
My infographic is set up in a flow chart style design to bring the audience on a journey through how much gasoline the U.S. consumes in a year and how much CO2 is produced as a result of the consumption. Since my Capstone Lab was based on biofuel I thought it made perfect sense to do my infographic on gasoline.
After conducting my scientific investigation I was able to learn about the building blocks of the fuel I was creating. I learned how each ingredient was mixed was mixed together and the bonds that were formed to make the final biodiesel. For my particular scientific investigation, the big idea that I took away was how specific ingredients, that cannot be directly used as fuel, can be mixed and come together and form a viable alternative for fuel.
The research I did for the creation of my infographic was very informative in an overwhelming manner. I was astounded by the amount of gasoline that the United States uses and how much CO2 is emitted as a result of gasoline's consumption. Though I did not do any research pertaining to the rate gasoline depletion across the globe I was still surprised about the amount we consumed based on my knowledge of how much only one person uses.
My infographic is set up in a flow chart style design to bring the audience on a journey through how much gasoline the U.S. consumes in a year and how much CO2 is produced as a result of the consumption. Since my Capstone Lab was based on biofuel I thought it made perfect sense to do my infographic on gasoline.
Elevator Pitch
This new and upcoming material that is much lighter and much stronger than any other material today will help to bring forth more efficient vehicles, stronger therefore longer lasting buildings and bridges, and even the possibility of space elevators. I am talking about carbon nanotubes. Carbon nanotubes, or CNTs for short, are the strongest and stiffest material ever discovered. They are hollow cylinders made up of one-atom-thick walls of graphene that have a diameter that can range from <1nm to 50nm and a length to diameter ratio of up to 132,000,000:1. Being 28 times stronger than steel, an individual CNT shell can have a tensile strength of up to 15,000,000 pounds per square inch. Applications for CNTs include conductive plastics, structural composite materials, gas storage, nano- and micro-electronics, radar absorbing coating, ultra capacitors, batteries with improved lifetime, and extra strong fibers. Their unique properties make the possibilities endless. Your generous contribution for funding further research and development of carbon nanotubes will make a massive technological impact on our society.
Materials Project
Carbon Nanotubes: Breaking the Barrier of Weak
Contemporary fibers and materials are beginning to become inadequate, not strong enough or light enough, for current and future scientific endeavors; with having a phenomenal strength, stiffness and tenacity as well as very high thermal and electrical conductivity, carbon nanotubes are the answer. Carbon nanotubes (CNTs) are hollow cylinders made up of one-atom-thick walls of graphene. Graphene is a sheet of carbon atoms bonded together in a hexagonal arrangement (a honeycomb.) A CNTs’ diameter can range from <1nm to 50nm and they have a length to diameter ratio of up to 132,000,000:1.
Applications for CNTs include conductive plastics, structural composite materials, gas storage, nano- and micro-electronics, radar absorbing coating, ultra capacitors, batteries with improved lifetime, and extra strong fibers. Their unique properties make the possibilities endless. The incredibly high strength of CNTs has been the most popular topic according to a group of scientists on phys.org in an article containing information on a multitude of tests on the strength and flexibility of CNTs. A material that is much lighter and much stronger than any other material today will help to bring forth more efficient vehicles, stronger therefore longer lasting buildings and bridges, and even the possibility of space elevators.
CNTs are the strongest and stiffest materials ever discovered. The reason for this tremendous strength is the covalent bonds between the carbon atoms that make up the graphene in the shape of a nanotube. This combination along with the honeycomb shaped walls (the honeycomb structure allows the greatest strength to weight ratio in terms of geometric proportion) of the nanotube is what gives CNTs strength much greater than any other material on Earth. A CNT made up of multiple walls of graphene was tested for strength in the year 2000 and came up to have a tensile strength of 9,100,000 psi. Later in 2008, another test was conducted which revealed that an individual CNT shell could have a tensile strength of up to 15,000,000 psi. A current and vastly utilized material called Kevlar, a material five times stronger than steel, only has a tensile strength of 525,000 psi.
The carbon atoms within layers of graphene are arranged in three different configurations, when looking at a nanotube, with each configuration having a different property. There is a zigzag arrangement, a chiral arrangement, and an armchair arrangement. If one were to conceptualize the structure of a CNT with a zigzag arrangement a square sheet of graphene would be folded into a cylinder with the two edges’ hexagon shapes matching up perfectly to form a clean structure. For conceptualizing an armchair arrangement, the folding of the sheet of graphene would be perpendicular to that of a zigzag and would form a different pattern on each end of the nanotube. A chiral arrangement would be if the graphene sheet were folded diagonally, the edges of the sheet may or may not match up and the ends of the tube would be ovular instead of circular. The properties of each arrangement vary from different strengths to conductivity.
CNTs were first completely discovered and synthesized in 1993 by scientists Sumio Iijimia and Donald Bethune (though the idea and possible analysis of CNTs have been happening since the 1950s.) Through a collaborative effort Iijimia and Bethune were able to produce a single-walled CNT by using transition metal catalysts. A carbothermal reduction of silica to silicon was used through the catalysts to build a carbon nanostructure formation. Today, the methods behind the growth of CNTs are controversial based on that some methods only grow them accurately on occasion and most of the methods are quite insufficient for the need of the current society. Due to the fact that they are universal and will not work in some cases as CNTs are extremely difficult to grow and maintain.
Contemporary fibers and materials are beginning to become inadequate, not strong enough or light enough, for current and future scientific endeavors; with having a phenomenal strength, stiffness and tenacity as well as very high thermal and electrical conductivity, carbon nanotubes are the answer. Carbon nanotubes (CNTs) are hollow cylinders made up of one-atom-thick walls of graphene. Graphene is a sheet of carbon atoms bonded together in a hexagonal arrangement (a honeycomb.) A CNTs’ diameter can range from <1nm to 50nm and they have a length to diameter ratio of up to 132,000,000:1.
Applications for CNTs include conductive plastics, structural composite materials, gas storage, nano- and micro-electronics, radar absorbing coating, ultra capacitors, batteries with improved lifetime, and extra strong fibers. Their unique properties make the possibilities endless. The incredibly high strength of CNTs has been the most popular topic according to a group of scientists on phys.org in an article containing information on a multitude of tests on the strength and flexibility of CNTs. A material that is much lighter and much stronger than any other material today will help to bring forth more efficient vehicles, stronger therefore longer lasting buildings and bridges, and even the possibility of space elevators.
CNTs are the strongest and stiffest materials ever discovered. The reason for this tremendous strength is the covalent bonds between the carbon atoms that make up the graphene in the shape of a nanotube. This combination along with the honeycomb shaped walls (the honeycomb structure allows the greatest strength to weight ratio in terms of geometric proportion) of the nanotube is what gives CNTs strength much greater than any other material on Earth. A CNT made up of multiple walls of graphene was tested for strength in the year 2000 and came up to have a tensile strength of 9,100,000 psi. Later in 2008, another test was conducted which revealed that an individual CNT shell could have a tensile strength of up to 15,000,000 psi. A current and vastly utilized material called Kevlar, a material five times stronger than steel, only has a tensile strength of 525,000 psi.
The carbon atoms within layers of graphene are arranged in three different configurations, when looking at a nanotube, with each configuration having a different property. There is a zigzag arrangement, a chiral arrangement, and an armchair arrangement. If one were to conceptualize the structure of a CNT with a zigzag arrangement a square sheet of graphene would be folded into a cylinder with the two edges’ hexagon shapes matching up perfectly to form a clean structure. For conceptualizing an armchair arrangement, the folding of the sheet of graphene would be perpendicular to that of a zigzag and would form a different pattern on each end of the nanotube. A chiral arrangement would be if the graphene sheet were folded diagonally, the edges of the sheet may or may not match up and the ends of the tube would be ovular instead of circular. The properties of each arrangement vary from different strengths to conductivity.
CNTs were first completely discovered and synthesized in 1993 by scientists Sumio Iijimia and Donald Bethune (though the idea and possible analysis of CNTs have been happening since the 1950s.) Through a collaborative effort Iijimia and Bethune were able to produce a single-walled CNT by using transition metal catalysts. A carbothermal reduction of silica to silicon was used through the catalysts to build a carbon nanostructure formation. Today, the methods behind the growth of CNTs are controversial based on that some methods only grow them accurately on occasion and most of the methods are quite insufficient for the need of the current society. Due to the fact that they are universal and will not work in some cases as CNTs are extremely difficult to grow and maintain.
Project Reflection
As the human civilization continues to expand and progress we persist at making new discoveries in the field of matter. Today, we live in a world driven by incredibly advanced technology, all the things we can do with technology are possible because chemistry allows it to happen. In the past some technologies were completely possible and conceivable because chemistry allowed it, but us humans were not yet smart enough to create what we have now. Specific elements bonding together with other elements or compounds mixed in are what create what we had in the past, what we have now, and what we will have in the near and distant future. Though in the future, there will just be some things we will not be able to achieve because chemistry will not allow it.
Based on a substances' certain molecular structure will cause it to exhibit certain properties on the microscopic and macroscopic levels. For example, looking at salt (sodium chloride) at the molecular level it will have sodium ions ionically bonded to chlorine ions in a cubic arrangement; which in turn causes it to be in cubic form on the microscopic level. At the macroscopic level one will notice that certain elements will bond to other elements, this is due to how many protons, electrons, and neutrons the elements have, which is observable and/or predictable on the atomic and molecular level. Such as how metals usually bond to nonmetals to complete the Octet Rule, a rule saying that in order for two elements of a main group to bond with one another they must exchange electrons to have them add up to a total of eight on their valence shell; which makes a stable compound.
Based on a substances' certain molecular structure will cause it to exhibit certain properties on the microscopic and macroscopic levels. For example, looking at salt (sodium chloride) at the molecular level it will have sodium ions ionically bonded to chlorine ions in a cubic arrangement; which in turn causes it to be in cubic form on the microscopic level. At the macroscopic level one will notice that certain elements will bond to other elements, this is due to how many protons, electrons, and neutrons the elements have, which is observable and/or predictable on the atomic and molecular level. Such as how metals usually bond to nonmetals to complete the Octet Rule, a rule saying that in order for two elements of a main group to bond with one another they must exchange electrons to have them add up to a total of eight on their valence shell; which makes a stable compound.